There are many people in the gold world trying to fool you. One especially sneaky trick is to try and sell you gold that is hallmarked, but not in fact authentic. That is to say, it says something like "18K" and is anything but. Before you buy gold bars second-hand, you need a toolkit to make sure you're getting the value you're paying for.
There are a number of ways to avoid getting defrauded. The things is that not all are equal. In fact, some of the best one are extremely impractical for the average person. How so? Well, let's start with the absolute best way to test gold: the fire assay. According to Republic Metals, an LBMA Good Delivery Refiner, "in most instances melting is required." A well-performed fire assay requires a large and well-trained team, in addition to significant capital investment.
Obviously, melting down gold you are buying would be hard enough, let alone testing it. So what's to do? Well, even the largest refiners start with something called an x-ray fluorescence or XRF for short. An XRF machine can costs anywhere from $15,000 to $100,000+.
Still, at Global Bullion team, like many large dealers, we know to take this test with a grain of salt. You see, the XRF tells you exactly what metals you're looking at on an elemental scale. However, its penetration depth is measured in microns and as such is really a surface analysis. One solution is to melt the gold and test it at various point. Is that something you're ready for?
Learning How to Test Gold With Acid Gives You a Toolkit Bigger Than a Magnet
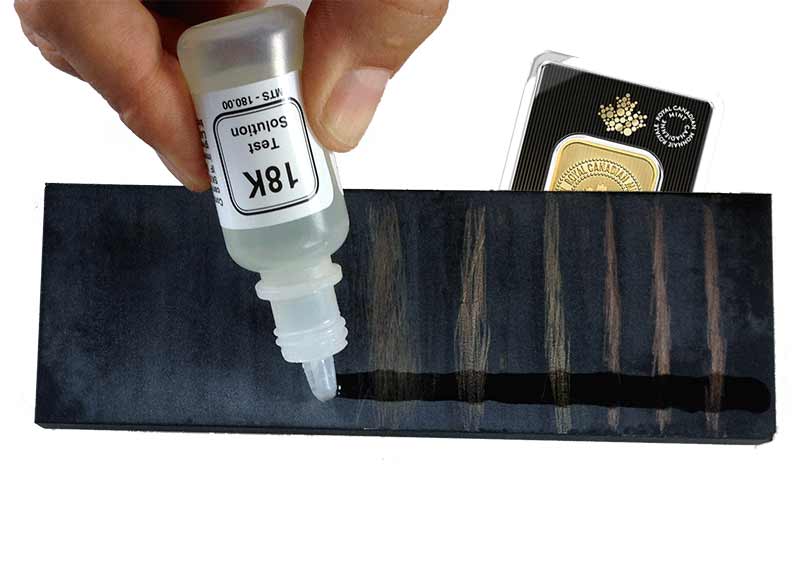
The acid test has a number of advantages. First, it's cheap: you can buy a gold acid-testing tool-kit on Amazon Canada from about CAD $20. Second, it's highly portable. Unlike a heavy and delicate XRF machine, you can throw an acid-test kit in a knapsack. Third, it's reasonably accurate at authenticating gold.
But if knowing how to test gold with acid is so good, why isn't everyone using it? First, it only tests the surface. Most experienced users will file into questionable specimens to test the inside of the gold. Second, the acid test is limited in accuracy to rough karat-counts: 10k, 14k, 18k, etc. The acid is pre-made to test for a certain karat and we wouldn't recommend using it for much else. In other words, watch out for false positives.
Finally, the third risk is that while you don't need an x-ray safety course, you need to watch not to make contact between acid or residue and your skin. It can hurt a little and leaves awkward yellow stains. Having said the above, you're probably wondering how to test gold with acid in real life. What does the step-by-step process look like? Well, lucky for you it's easy.
How to Test Gold with Acid, Step-By-Step:
Step 1: Unpack
Take out the testing acids, testing stone, some ammonia (or Windex) for cleanup, and a put on a pair of latex gloves.
Step 2: Inspect
Inspect each piece of gold, looking for karat marks on jewellery and fineness on bars. A gold bar from an LBMA good-delivery refinery must be at least 99.5% gold. Group them together.
Step 3: Scratch
Scratch (making a separate line) each piece gold from a given group against the stone. You will test these lines.
Step 4: Use the Acid
Apply a drop of the acid to each item's representative line. Watch for how much gold remains, as opposed to base metal dissolved by the acid.
Step 5: File it.
Concerned that your gold isn't living up to the hype? Take a tough metal file and scrape deep into it. Put the exposed area into several drops of acid for 30-60 seconds. See green or signifiant activity? Don't risk it.
Step 6: Make a Decision.
The acid test just told you roughly what % of gold is in front of you. Now you can make a much better decision on the risk of fraud. Remember to always be careful and if unsure, file some more.
Now That You Know How to Test Gold with Acid, How Does it Work?
Strictly speaking, there really aren't many scientifically validated ways to test gold. Moreover, there are very few ways to test gold easily and at a reasonable cost. That's where the chemistry comes in. Remember that scratch you made on the stone? You left a very fine residue of the metal on it. You are then experimenting on that residue to determine the composition of the whole. While it requires sacrificing a tiny amount of gold on the stone, it leaves the rest of the material intact (unlike melting it).
The acid test works for the same reason many people find gold pleasant to wear. It is extremely stable and won't react to anything but the strongest of compounds. Pre-determined strengths of of aqua regia (latin) with increasing amounts of nitric and hydrochloric acid will dissolve varying amounts of metal. The stronger the acid the gold withstands, the more pure it is. Equipped with an acid-test kit and knowledge, you should be a little more confident in buying precious metals, regardless of from friend or foe!